(Disclaimer: This article contains images that can affect you emotionally, readers discretion is advised)
 he medical industry had come a far way in the past 100 years, but sometimes the hunger for more profits can lead to devastating consequences. Do you know that type of medicine that is so efficient it could be used to treat a lot of medical problems? You don’t because it does not exist. If you purchase medicine without a prescription you should always check the instructions and never trust the internet for advice.
he medical industry had come a far way in the past 100 years, but sometimes the hunger for more profits can lead to devastating consequences. Do you know that type of medicine that is so efficient it could be used to treat a lot of medical problems? You don’t because it does not exist. If you purchase medicine without a prescription you should always check the instructions and never trust the internet for advice.
This is something a pharmaceutical company in the 1950s didn’t take into account and it lead to a whole generation of children being born disfigured. Selling medical products that have been produced for one intent and advertising them as if they can cure many different things was difficult to monitor back in the 1950s as there weren’t as many checks as there are today and laws weren’t very specific.
Thalidomide: the miracle medicine
It all started with Grünenthal, one of the biggest pharmaceutical companies around the world which originated in Germany. The company turns over $2 billion in revenue per year. The company was founded in 1946, right after World War II by a German family in Aachen. This was also the firm pharmaceutical company to introduce penicillin within the German market after the ban on penicillin was lifted.
At the start of the 1950s, the company started to develop a new drug intended to be used as a sedative or tranquilizer for humans. During the testing trial which only occurred on animals, the drug was deemed 100% harmless to humans. By the time Thalidomide reached the pharmacy counters in Germany, the drug was advertised that it could treat a wide range of other conditions such as colds, flu nausea, and (very important) morning sickness in pregnant women.
In July 1956, Thalidomide was licensed to be sold over the counter without the need for a doctor’s prescription. Other pharmaceutical companies around the world were attracted by the drug to the number of medical issues it was treating. Therefore by 1957, 14 different pharmaceutical companies bought the license from Chemie Grünenthal to produce and sell the same product in 46 countries around the world under at least 37 different trade names.
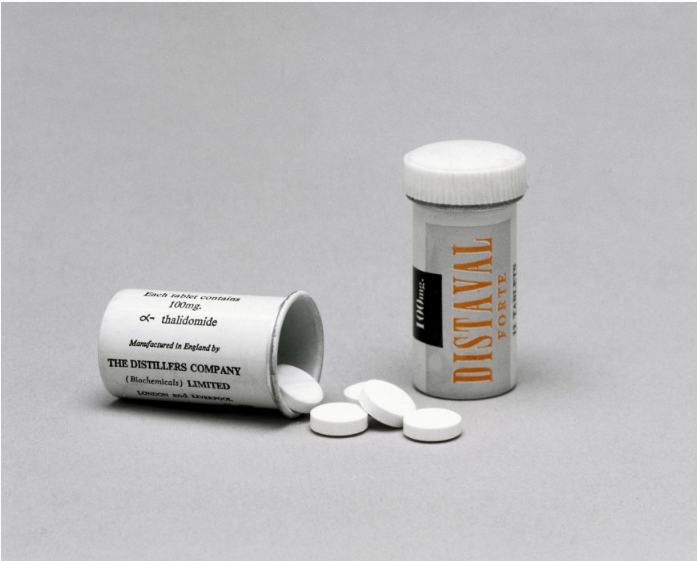
Within the United Kingdom, thalidomide was sold under the name Distaval (among many others) produced by the “The Distillers Company Ltd”. One very interesting aspect about Distaval specifically is that its description mentioned:
“Distaval can be given with complete safety to pregnant women and nursing mothers without adverse effect on mother or child.”
The drug was specifically focused on pregnant women as other drugs that would cure illnesses such as colds, flu, headaches, and so on were deemed to be stronger, therefore with a potential to affect the child. thalidomide was recommended by doctors around the world towards women that had colds, flu, or nausea which is something common in early pregnancy.
Interestingly enough, in the United States, thalidomide was rejected by the Food and Drug Administration, therefore it was never sold in the states. Several requests had been made by people towards the U.S. government to approve the drug, but they never did, and thank God for that. People were so desperate for the drug that they would fly out in countries that it was sold in order to procure it.
“Why is my child missing a limb?”
Was the question that many mothers around the world started asking in 1960 as they would give birth to children that were missing limbs or in some cases they weren’t even making it through their first few days due to the indescribable disfigurement they were born in. Here are some photos to show what some children were born like.

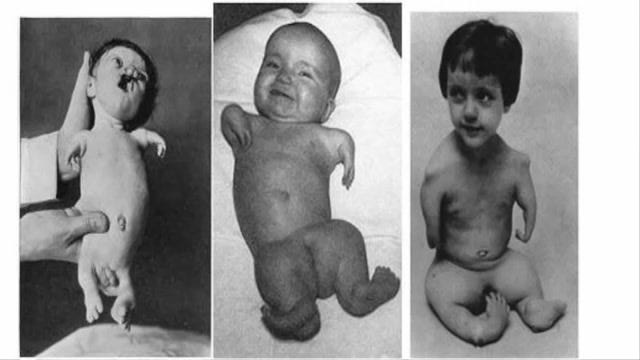
The photos you are seeing above are some of the worst cases, but in most common cases where the children of pregnant women had been affected by thalidomide, either their arms or legs would be missing. At first (1960) there were a small number of cases and medics blamed it on a multitude of things, but none of them actually looked at thalidomide as a potential cause. It is also important to mention that in all the trials that thalidomide went thru with human subjects, it was never tested on pregnant women.

By 1961, the number of children born with such malformations was on the rise and Doctors did start to look at the consumption of pregnant women that had their children affected. All of the women had the same thing in common, they all took thalidomide during their pregnancy. Until the world actually came to a clear conclusion that thalidomide was the cause of this it took 5 long years and this is due to all the different malformations and complications that the babies were born with.
Here is a list of the impacts that thalidomide may have on the fetus when taken by pregnant women:
- Bilateral limb atrophy (legs, arms or both)
- Phocomelia
- Bilateral limb absence (amelia)
- Missing fingers or toes
- Palmature of the fingers or toes
- Extra fingers or toes
- Total or partial hearing loss
- Partial or total vision loss
- Paralysis (usually facial muscles)
- Malformation of the digestive tube
- Malformation of the duodenum (most of the time lethal, before or not long after birth)
- Malformation or absence of the anus, vital organs injury (most of the time lethal, before or not long after birth)
- Death
What are the causes?
Even to this day, the cause by which thalidomide causes birth defects are unknown, but there are some theories that experts came up with over the years. From the papers that I have read, the most plausible one seems to be Takumi et al 2010 entitled “Identification of Primary Target of Thalidomide Teratogenicity”. Thalidomide exerts teratogenic effects by binding to cereblon and inhibiting associated enzymatic activity important for limb development. In other words, thalidomide interferes in the development of limbs within fetuses.
However, it is very difficult to define a specific cause because there are too many issues caused by thalidomide in these babies.
The first time the link between thalidomide and its impact on development was made public was in a letter published in The Lancet from an Australian doctor William McBride, in 1961. In November of 1961, Grünenthal had formally withdrawn the drug, however, in other countries, the drug was still produced and sold for a couple of more years.

Many distributors sued the pharmaceutical companies that produced thalidomide and many of the same companies sued Grünenthal for being at fault for all this. It is true that Grünenthal is mostly at fault, but in my opinion, it is also the government that failed to test the drug for all circumstances just like the FDA did in America.
Due to this drug, over 10,000 children had been born with malformations and half of them died in the first month of being born. Today, many of the western governments that are housing children affected by thalidomide are offering lifetime support, although some of the survivors say that these funds still don’t make up for the terrible medical disaster.
Avid Writer with invaluable knowledge of Humanity!
Upcoming historian with over 30 million views online.
“You make your own life.”





